
Nitrates and Nitrites in Drinking Water: What You Should Know
By Dan DeBaunShare
Nitrates and nitrites in water are critical contaminants that pose significant health risks to humans and ecosystems. These compounds, often originating from agricultural runoff and wastewater, can lead to serious conditions such as methemoglobinemia in infants and contribute to broader environmental degradation. Understanding their sources and impacts is essential – so let's learn more.
In this article, you'll learn:
- What are Nitrates and Nitrites in Drinking Water?
- Nitrate Pollution From Fertilizers
- Non-agricultural Sources of Nitrogen
- What is the Difference Between Nitrates and Nitrites?
- Are Nitrates in Water Bad For You?
- Effects of Nitrates in Drinking Water on Humans
- How to Test for Nitrates and Nitrites in Drinking Water
- How to Remove Nitrate and Nitrite from Drinking Water
- Understand Your Water Quality
What are Nitrates and Nitrites in Drinking Water?
Nitrates (NO₃⁻) and nitrites (NO₂⁻) are nitrogen-containing compounds commonly found in water sources. They are part of the nitrogen cycle and are introduced into water through various natural and anthropogenic activities. Here is an overview of what nitrates and nitrites are, their sources, and their significance.
Since the 1950s, farmland (and more recently lawns) have been soaked in nitrogen-rich fertilizers. As a result, nitrogen from fertilizers has been leaching into groundwater for decades. When these fertilizers leach into the surrounding natural water systems, harmful contamination results, and our drinking water is ultimately affected.
Though our bodies do not need nitrogen in this gaseous form, plants that we eat do. In fact, all living systems need nitrogen in some form since nitrogen is used to build many essential components such as proteins, DNA, RNA, and vitamins, as well as hormones and enzymes.
The plants we eat use simple nitrogen such as ammonium nitrate to make complex nitrates such as amino acids and nucleic acids. This latter form of nitrogen we can use. Plants work as ‘mediators’ to take unusable nitrogen and convert it to a usable form for us. However, this also means that some forms of nitrogen that may be good for plants can be toxic to us.
Nitrate Pollution From Fertilizers
You have no doubt noticed three numbers on a bag of fertilizer such as 20-5-10. These numbers relate to the percentage of nitrogen, phosphorus, and potassium in the fertilizer. This form of nitrogen is great for plants, but when it enters groundwater or runs off into streams and rivers, it throws the surrounding ecosystems out of balance, causing eutrophication and algal blooms, resulting in the deterioration of the ecosystem.
Nitrogen fertilizers are not the only sources of nitrate pollution in water. Animal manure, including livestock and poultry manure piles and nitrogen-rich manure used as fertilizer, is also a source of contamination. The good news is that some responsible agricultural producers such as Yakima Dairies are taking steps to reduce nitrate contamination in groundwater.
Non-agricultural Sources of Nitrogen
In addition to fertilizers and manure, nitrate-containing wastes are produced by many industrial processes including paper and munitions manufacturing. Burning of fossil fuels in power plants, motor vehicles, and all internal combustion engines results in the production of nitric acid and ammonia as air pollution. This air pollution can eventually enter our waterways when it takes the form of acid rain and falls back to the earth.
What is the Difference Between Nitrates and Nitrites?
Nitrates and nitrites are both nitrogen-containing compounds, but they have distinct chemical structures, properties, and roles in environmental and biological systems. Nitrates (NO₃⁻) consist of one nitrogen atom bonded to three oxygen atoms, while nitrites (NO₂⁻) consist of one nitrogen atom bonded to two oxygen atoms. While nitrates tend to be more stable than nitrites, both are highly soluble in water.
Nitrates occur naturally in soil and are formed as a result of the nitrogen cycle when decaying plant matter is broken down by bacteria. However, their levels can be increased by contributions from anthropogenic sources such as fertilizers, animal waste, wastewater, and industrial processes. Nitrates are also used in the food industry as preservatives and color fixatives in cured meats and in some pharmaceutical and medical treatments.
Nitrites, on the other hand, are products of the nitrogen cycle, formed by the reduction of nitrates by bacteria. Nitrite is used in the food industry as a preservative to inhibit bacterial growth and to preserve the color and flavor of meat products. It is also used in various industrial chemical processes, including the production of dyes and corrosion inhibitors.
Are Nitrates in Water Bad For You?
While nitrates are generally considered less harmful than nitrates, they can be converted to nitrites in the body, which pose a greater health risk.
Nitrites can interfere with the oxygen-carrying capacity of blood, leading to methemoglobinemia, especially in infants (blue baby syndrome). They can also form nitrosamines, which are potential carcinogens, especially in the presence of amines in the stomach. Studies have shown that exposure to nitrates in drinking water increases the risk of colorectal cancer.
As exposure to nitrates and nitrites in particular can potentially cause adverse health effects, they are both regulated in drinking water by agencies such as the US Environmental Protection Agency (EPA) and World Health Organization (WHO). The EPA maximum contaminant level for nitrites in drinking water is more stringent at 1 part per million (1 mg/L) compared to that of nitrates, which is set at 10 parts per million (10 mg/L).
Effects of Nitrates in Drinking Water on Humans
Methemoglobinemia, also called “blue baby,” is a serious health issue caused largely by high nitrate levels in drinking water. These high nitrate levels interrupt the normal body processes of some infants, especially those under 6 months of age. Nitrate becomes toxic when it is transformed to nitrite, a process that can occur in the stomach or the saliva of the infant. Babies are especially susceptible because their stomach juices are weaker and therefore are more likely to permit the production of nitrate-reducing bacteria in their stomachs.
Nitrite in the blood combines with hemoglobin to form methemoglobin. This reduces the capability of the blood to carry oxygen around the body. The result is the “blue” coloration of the baby’s skin. The skin turns blue because the infant is being asphyxiated as oxygen cannot be transported by the blood. If this is observed, medical help should be sought immediately.
Boiling the water will not reduce the nitrate concentration; in fact, it actually increases the concentration by evaporating the water and thus concentrating the pollutants. Water that is high in nitrates should not be used for preparing infant formula or any food that could be consumed by a baby. Though fatalities are rare, methemoglobinemia can insidiously show no symptoms while it is affecting child or animal development.
How to Test for Nitrates and Nitrites in Drinking Water
As exposure to high levels of nitrates and nitrites can have serious health implications, testing for these compounds is important for ensuring the safety and quality of drinking water. You can test your drinking water for nitrates and nitrites at home using a home test kit or digital meters. Alternatively, you can collect a sample and send it to a laboratory for testing.
Home test kits consisting of either test strips that change color when dipped into a water sample, or colorimetric test kits that involve adding a reagent to the water sample, which are then compared against a color chart provided with the kit, offer a cheap and easy method of testing for nitrates and nitrites at home.
Digital nitrate- and nitrite meters, which are calibrated for nitrate or nitrite respectively, also offer a simple method for testing the levels of these compounds in drinking water at home. Once the meters have been calibrated according to the manufacturer’s instructions, just insert the probe into the water sample and wait for the reading to stabilize before recording the displayed level.
Alternatively, you can send a water sample to a laboratory that offers professional water testing services. Collect a water sample using a sterile container provided by the lab. Ensure that you follow the lab’s instructions for sample collection, preservation, and shipping. Submit the sample to the lab for analysis, and once they have analyzed the sample, you will receive a detailed report with precise nitrate and nitrite levels.
How to Remove Nitrate and Nitrite from Drinking Water
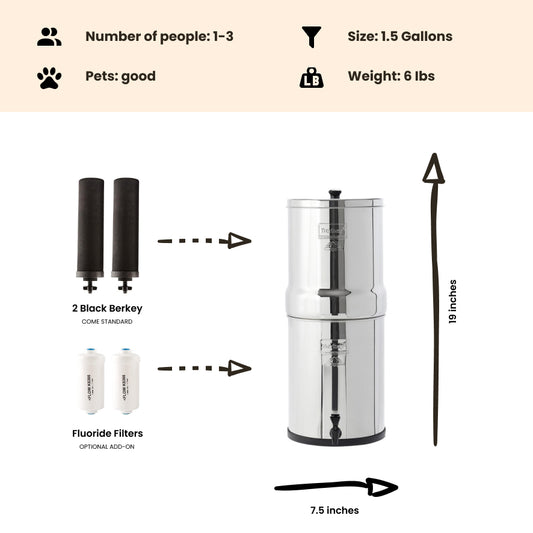
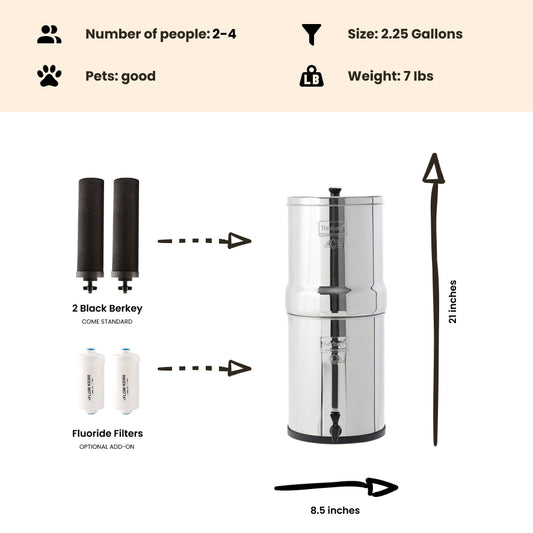
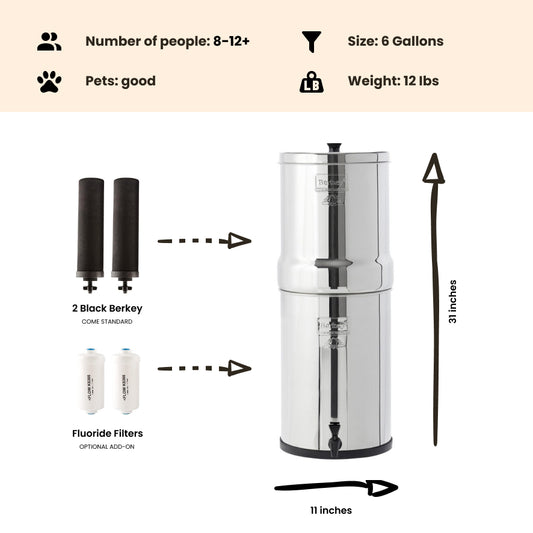
If levels of nitrite in your drinking water exceed the strict safety standard of 1 part per million (1 mg/L), a Berkey water treatment system fitted with Black Berkey filter elements can remove more than 95% of the more harmful nitrites from your water supply to bring concentrations within acceptable safety limits. However, if you have unacceptable levels of nitrates (concentrations exceeding 10 parts per million / 10mg/L) in your drinking water, you will need to consider investing in a reverse osmosis filter.
Understand Your Water Quality
Nitrates and nitrites in water are important indicators of water quality, originating from agricultural, industrial, and natural sources. Both compounds pose significant health risks and environmental concerns, particularly in high concentrations.
Monitoring and regulation, combined with effective treatment and mitigation strategies, are essential to manage their levels and protect public health and ecosystems. Nitrates and nitrites are just two of hundreds of common drinking water contaminants. So we highly encourage you to learn about other common drinking water contaminants that can affect the quality of your drinking water!
Nitrates and Nitrites in Drinking Water: FAQ
What are nitrates and nitrites?
Nitrates and nitrites are nitrogen-oxygen chemical compounds that play significant roles in various environmental and biological processes. While they are essential for plant growth and food preservation, their presence in high levels in drinking water can pose significant health risks.
What is the difference between nitrates and nitrites?
Despite their similar names and chemical compositions, nitrates and nitrites differ in their structures, sources, and effects. Nitrates, which consist of one nitrogen atom and three oxygen atoms, are more stable and commonly found in nature and agricultural products, while nitrites, which consist of one nitrogen atom and two oxygen atoms, are more reactive and used mainly as food preservatives. Both compounds are regulated to minimize their potential adverse effects on human health and the environment.
What causes high nitrates in drinking water?
High nitrates in drinking water are primarily caused by agricultural activities, wastewater and sewage, industrial sources, natural processes, urban runoff, and poor land management practices.
What happens if you drink water with nitrates and nitrites?
Drinking water contaminated with high levels of nitrates and nitrites can cause a range of health problems and potential health risks, including methemoglobinemia (especially in infants), potential cancer risk due to nitrosamine formation, and reproductive complications. It is essential to regularly monitor your water quality to ensure it adheres to regulatory standards, and to implement effective treatment and preventive measures to mitigate these risks to ensure your water is safe to drink.
-
Regular price $234.00 USDRegular priceUnit price / per
-
Regular price $327.00 USDRegular priceUnit price / per
-
Regular price From $367.00 USDRegular priceUnit price / per
-
Regular price From $408.01 USDRegular priceUnit price / per
-
Regular price From $451.00 USDRegular priceUnit price / per
-
Regular price From $478.00 USDRegular priceUnit price / per
-
Regular price $332.50 USDRegular priceUnit price / per
$350.00 USDSale price $332.50 USDSale

Dan DeBaun is the owner and operator of Big Berkey Water Filters. Prior to Berkey, Dan was an asset manager for a major telecommunications company. He graduated from Rutgers with an undergraduate degree in industrial engineering, followed by an MBA in finance from Rutgers as well. Dan enjoys biohacking, exercising, meditation, beach life, and spending time with family and friends.
~ The Owner of Big Berkey Water Filters













Nitrates can be devastating to our health. I recommended a berkey water filter to all! No exceptions!